Science
- Home
- Science
- Excellence
- Surface chemistry and nanotechnology
-
Excellence
- Advanced materials and technologies
- Surface chemistry and nanotechnology
- Photonic and Laser Technology
- Metrology and measurement technology
- Environmental Research and energetics
- Organic materials and nanoenginering
- Electrochemical energy conversion technologies
- Research of the structure and composition of solid materials
- Long-Term Programs
- Scientific publications
- Conferences
- Projects
Surface chemistry and nanotechnology
The scientists of Nanostructures laboratory of the Department of Electrochemical Material Science during 2019-2023 years investigated the synthesis of new nanostructured electrocatalysts for hydrogen evolution from water. The results obtained were published in eight Q1 and Q2 journals, reported in several international conferences, and two defended doctoral dissertations. It has been established that inserted histidine amino acid fragments in the 1T/2H-MoS2 nanoplatelet structures significantly increased activity and stability of hydrogen evolution reaction (HER). Just 6 mg/m2 of Pt enough for decoration of MoS2 layers to achieve HER activity as at the Pt bulk. Red-fluorescent magnetic nanoparticles have been synthesized in this Laboratory for the first time. These nanoparticles recently are studied in the National Cancer institute for detection of cancer cells. In this laboratory, nonstoichiometric TiOx nanoparticles have been also synthesized on the Ti surface for the first time. Their nontypical optical properties have been applied for fabrication gas sensors, referred in several papers, doctoral dissertation and international conferences.
The activities of the Department of Catalysis are directly focused on the research and development of sustainable and low-carbon energy conversion and storage technologies – electrochemical hydrogen production as fuel via water splitting, low-temperature polymer membrane fuel cells, metal-air batteries, and supercapacitors. The research is focused on the design and development of high-performance multifunctional catalytic materials using electrochemical, electroless metal plating, microwave, and hydrothermal synthesis. Noble and non-noble transition metal and their alloy coatings with various structures, compositions, and extremely large electrochemically active surface areas, deposited on various conductive and non-conductive flexible substrates (metal and their foams, polymers, such as polyethylene (PE), polypropylene (PP), polyvinyl chloride, polyimides, polyetherimides), carbon, graphene powder, N-doped carbon from biowaste, graphitic carbon nitride, silicon carbide or quartz particles with enhanced activity towards the oxidation of various fuels (sodium borohydride, methanol, ethanol, hydrazine, formic acid, etc.), hydrogen and oxygen evolution reactions (HER and OER), oxygen reduction reaction (ORR) have been developed. The scientists of the Department of Catalysis have experience in the research on the electrocatalytic activity of the created materials for various catalytic applications using electrochemical methods, such as cyclic voltammetry, chrono-techniques, rotating disk or ring-disk electrode (RDE and RRDE), electrochemical impedance spectroscopy (EIS) methods.
Another important topic is the development of commercially viable and effective bio-based carbon materials, which are the potential candidates to be employed in low-carbon energy technologies such as ultracapacitors, metal-air batteries, and low-temperature polymer membrane fuel cells. The created innovative, high-performance, highly conductive, electrocatalytically active, durable, cost-effective, and high surface area nanocarbon materials with controlled pore size distribution from the waste of the wood industry, biorefineries, as well as the pulp and paper industry can be applied as supercapacitor and batteries electrodes and as catalysts for fuel cells.
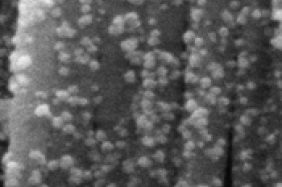
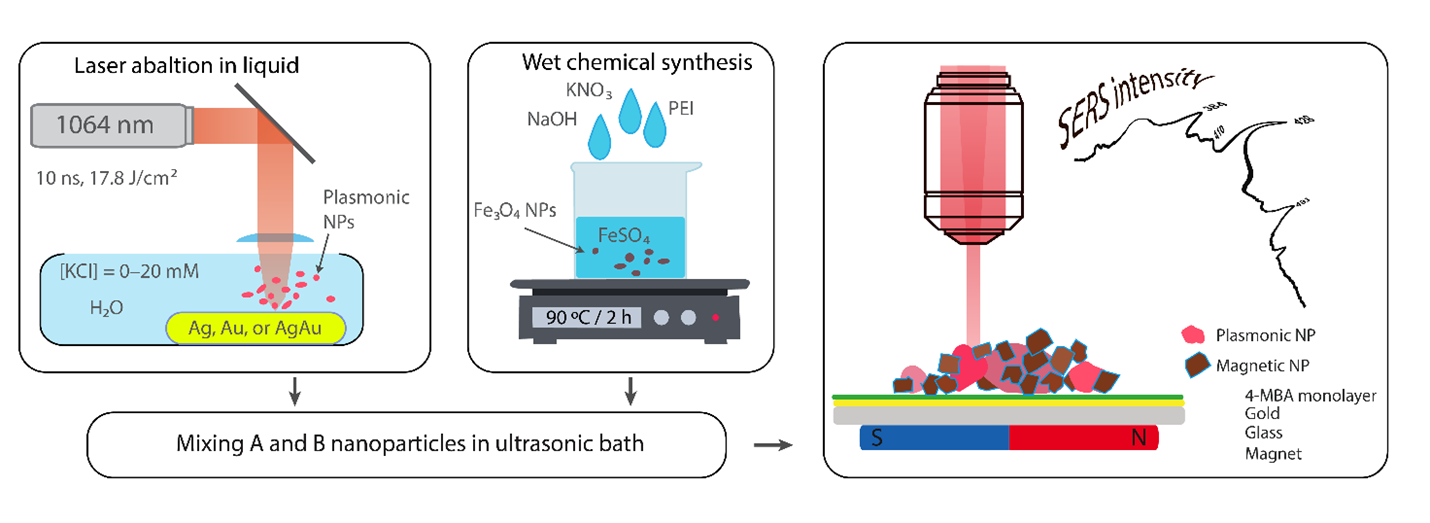
Scientists from the Spectroelectrochemistry laboratory of the Department of Organic Chemistry in collaboration with colleagues from the Laser Technology Department have developed novel synthesis methods of magneto-plasmonic nanoparticles based on combination of laser ablation and chemical synthesis approaches. Such nanoparticles exhibit both magnetic and plasmonic properties and can be widely used in spectroscopy, nanotechnology, and biomedicine. In collaboration with scientists from Vilnius University Chemical Physics Institute the effectiveness of such nanoparticles in distinguishing cancerous from healthy tissue using surface-enhanced Raman spectroscopy (SERS) has been demonstrated, spectral markers of cancer have been identified. Spectroelectroochemistry laboratory scientists continuously working on development of surface vibrational spectroscopy methods: SERS, surface enhanced infrared absorption spectroscopy (SEIRAS), and sum frequency generation spectroscopy. Raman spectrometer is capable of operating with 7 different laser sources, thus covering a wide range from the UV to the IR, allowing the observation of different nanomaterial parts of the system under study. An electrochemical cell added to the system allows the measurement of the materials on the surface and the processes taking place by changing the surface potential.
Optical biological sensors are one of the leading topics in the Department of Nanotechnology. The spectroscopic ellipsometry enhanced with surface plasmon resonance techniques and supplemented with quartz crystal microbalances is used for this purpose. Also, in the Department of Nanotechnology, electrochemical sensors are being researched as research objects using thin layers of electrically conductive polymers on electrodes. Such thin polymer layers are used in studies of electrochromic properties, applications of molecular imprinting polymers in producing electrochemical sensors to determine small molecular compounds in solutions, etc.
- Nanostructures laboratory (Head Dr. A. Jagminas)
- Department of Catalysis (Head dr. L. Tamašauskaitė-Tamašiūnaitė)
- Laboratory of Spectroelectrochemistry (Head prof. G. Niaura)
- Department of Nanotechnology (Head. habil. dr. Arūnas Ramanavičius)